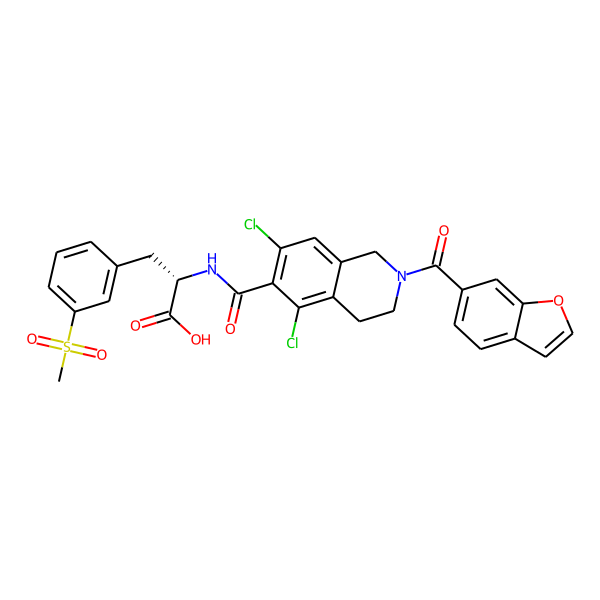
Lifitegrast
Function
DrugBank ID:
Description:
Lifitegrast is a FDA approved drug for the treatment of keratoconjunctivitis sicca (dry eye syndrome). It is a tetrahydroisoquinoline derivative and lymphocyte function-associated antigen-1 ( LFA-1) antagonist that was discovered through the rational design process. The ophthalmic solution was approved in July, 2016 under the trade name Xiidra. It has shown to protect the corneal surface and alleviate the symptoms of dry eye syndrome with fast onset of action and well tolerated profile in both local and systemic setting. [DrugBank]
Targets:
Integrin alpha-L (Humans) [DrugBank]
Pharmacodynamics:
Lifitegrast addresses both the symptoms and the resulting ocular surface damage by interfering with ocular inflammatory cycle 2. Lifitegrast is a lymphocyte function–associated antigen-1 antagonist through direct competitive antagonism and sequentially inhibits the T-cell recruitment, activation, and proinflammatory cytokine release associated with dry eye syndrome 4. [DrugBank]
Structures
Docking in target protein
Off-target analysis based on ligand similarity (Homo sapiens)
Step 1 - Target prediction for Lifitegrast: SwissTargetPrediction
Tips: Click on the link to jump to the 'SwissTargetPrediction' webserver. Select the species of 'Homo sapiens', and then paste the SMILES of Lifitegrast in the SMILES input box.
Step 2 - Blind docking for Lifitegrast: CB-Dock
Tips: Click on the link to jump to the 'CB-Dock' webserver. Upload the structure file of target predicted by 'SwissTargetPrediction' and the 2D/3D structure file of Lifitegrast to perform blind docking.